 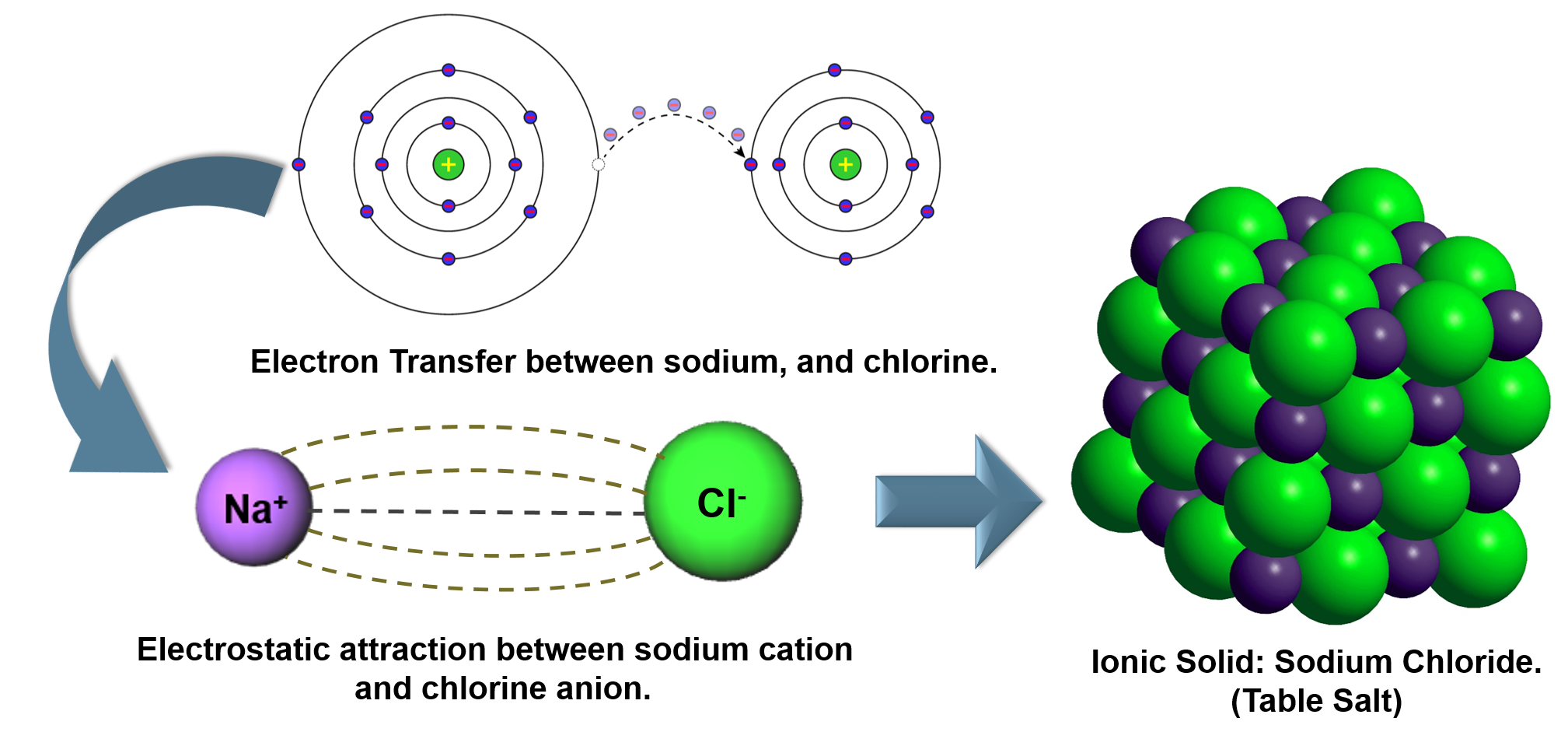 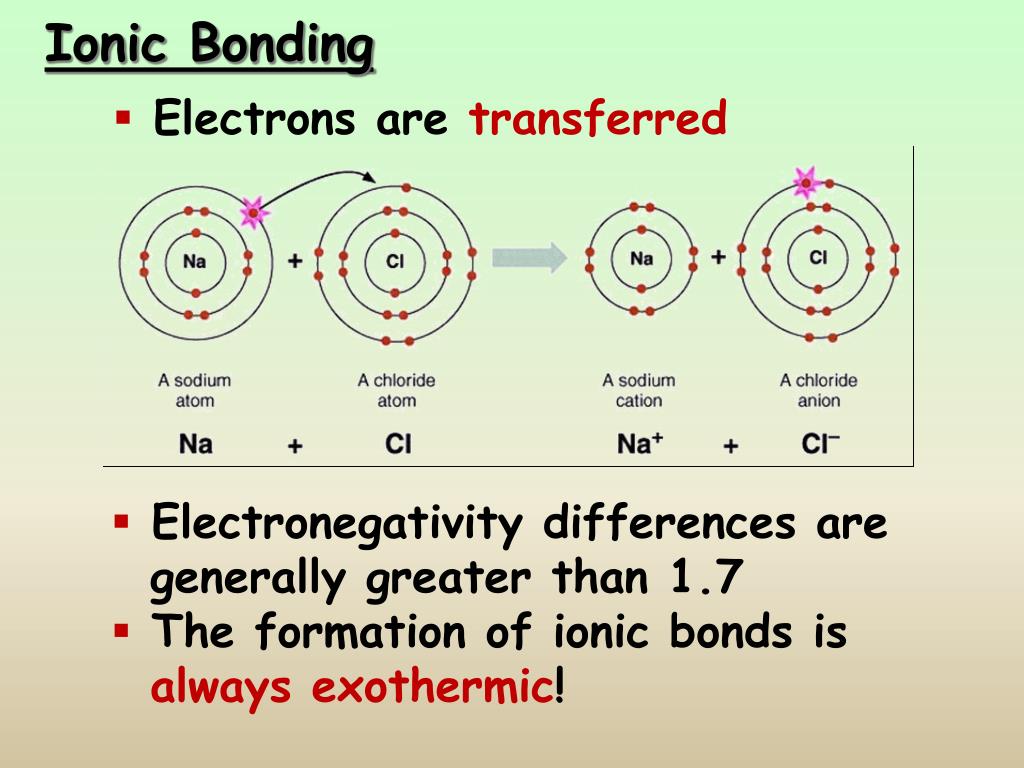
Examples of ionic compounds are NaCl, MgO and CaF 2. The metal gives electrons to the non-metal so that both achieve a noble gas configuration. Ionic compounds are formed from metal cations and non-metal anions. Thus sodium chloride is formed.Īnother example of ionic bond is the formation of magnesium chloride by transferring two electrons from a magnesium atom to two chlorine atoms. Ionic bonding tends to occur between electropositive metals and electronegative non-metals. Ionic bonding is the electrostatic attraction between oppositely charged ions. An electrostatic force holds these to atoms to together in a crystallographic lattice. Sodium losses an electron to get positive charge and chlorine accepts that electron to achieve a negative charge. As a result an electrostatic force occurs between these two oppositely and equally charged atoms to form an ionic bond. Collisions: Ionic Bonding Key Vocabulary Ion: an atom or group of atoms that has a positive or negative electric charge as a result of having lost or gained. The number of electrons it accepts, is the number of negative charge it gains. On other hand to fulfill the octet rule, non metals accepts the electrons and charged negatively. Electrons transfer from sodium atoms to adjacent chlorine atoms, since the valence electrons in sodium are loosely bound and chlorine has a large electron affinity. The number of electrons, a metal loses is the number of positive charge it achieves to form the ionic bond. A prototypical example is the sodium chloride crystal, as we discussed earlier. Thus it likes to give away these electron to achieve the noble gas configuration and satisfy the octet rule. Ionic bond occurs between metals and non-metals because the metals have only few electrons in its outermost shell. Many other types of bonding exist as well. A van der Waals bond occurs due to the attraction of charge-polarized molecules and is considerably weaker than ionic or covalent bonds. A covalent bond occurs when two or more atoms share electrons. Ionic bonding refers to the complete transfer of valence electrons. An ionic bond forms when an electron transfers from one atom to another. The metal that gives electrons is called donor and the non-metal that accepts electrons is called acceptor. Ionic bonds are formed by the combination of positive and negative ions the combination of these ions form in numerical combinations that generate a neutral (. Ionic bond refers to a type of chemical bond which generates two oppositely charged ions. As for example: metals such as sodium losses electrons to to become positive ion, whereas non-metal such as chlorine accepts electrons to become a negative ion. The energy of the electrostatic attraction ( E ), a measure of the force’s strength, is inversely proportional to the internuclear distance between the charged particles ( r ): (4.1. This is because they all have a full outer shell of electrons.Ionic bond is a kind of chemical bond which involves an electrostatic attraction between two oppositely charged ions because of the complete transfer of valence electrons between them. Ionic bonds are formed when positively and negatively charged ions are held together by electrostatic forces. They rarely react with other elements, be it water, oxygen, or metals. Group 8 elements, which you also know as noble gases, are known for being inert. Saturated Unsaturated and Supersaturated.Reaction Quotient and Le Chatelier's Principle.Prediction of Element Properties Based on Periodic Trends.Molecular Structures of Acids and Bases.The resulting compound is called an ionic compound. Ion and Atom Photoelectron Spectroscopy Ionic bonding When metals react with non-metals, electrons are transferred from the metal atoms to the non-metal atoms, forming ions.Elemental Composition of Pure Substances.Application of Le Chatelier's Principle In particular, the probe explores the extent to which students see ionic bonding in sodium chloride as a molecular phenomena, with discrete NaCl ion pairs which. Ionic bonding is a form of chemical connection in which one atom loses valence electrons and gains them from another.Intramolecular Force and Potential Energy.Thus by the formula, V 6 3 hydrogen atoms are bonded to oxygen, so the number of the monovalent atoms (M) 3 As this is a cationic molecule thus, C 1 So, H ½ 6 + 3 1 4 Thus we can say hybridization of H3O+ is Sp3. Variable Oxidation State of Transition Elements In hydronium ion, the central atom is oxygen and it has 6 valence electrons.Unlike covalent compounds, there is no such thing as a molecule of an ionic compound. The most common example of an ionic compound is sodium chloride NaCl, better known as table salt. /ionic-bond-58fd4ea73df78ca1590682ad.jpg)
Transition Metal Ions in Aqueous Solution The strong electrostatic attraction between adjacent cations and anions is known as an ionic bond.Single and Double Replacement Reactions. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
